Introduction
Approximately 60%-80% of patients with acute myeloid leukemia (AML) who achieve complete remission eventually relapse after conventional chemotherapy and have poor prognoses despite the recent advances of novel anticancer agents. Targeting topoisomerase 2 (Top2) is important to treat patients with AML, as anthracycline is widely used in general practice. However, anthracycline-induced cardiotoxicity or anthracycline resistance due to P-glycoproteinoverexpression often becomes major obstacles to successful treatment. Therefore, continuing development of more effective novel treatments for AML is necessary. Quinolones, antibacterials that attack DNA gyrase and topoisomerase IV (functional analog of mammalian Top2), possess a broad spectrum chemotherapeutic activities including anti-leukemic effects (e.g. vosaroxin). In this study, we developed and characterized a novel quinolone that demonstrates promising activities against human AML as a single agent and in combination with other agents.
Methods and Results
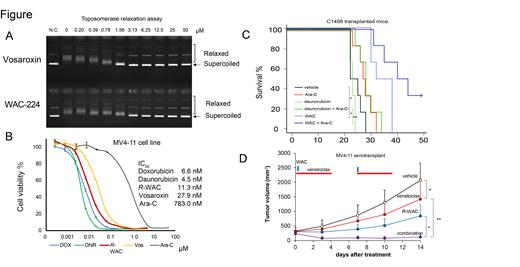
We developed 5-methyl-7-{3-[(oxan-2-ylmethyl) carbamoyl] azetidin-1-yl}-4-oxo-1-(1,3-thiazol-2-yl)-1,4-dihydro-1,8-naphthyridine-3-carboxylic acid [racemic WAC-224 (WAC) or (R)-WAC-224 (R-WAC), which is a novel anticancer quinolone, targeting Top 2. WAC inhibited Top2α comparable with vosaroxin (a known Top2 inhibitor) in a topoisomerase relaxation assay. IC 50 of vosaroxin and WAC were 1.10 μM and 2.26 μM, respectively. R-WAC significantly inhibited the human AML cell line proliferation (MV4-11, HL60, and KG1a), which was comparable to daunorubicin and cytarabine, in a manner not affected by P-glycoprotein overexpression. WAC also elicited cell apoptosis against MV4-11 cells comparable with vosaroxin. Whereas, WAC did neither increase serum troponin-T nor decrease the crypt numbers in the small intestine, indicating WAC was less toxic than doxorubicin. R-WAC monotherapy demonstrated prolonged survival in the AML mice model and inhibited tumor growth in the MV4-11 xenograft mice model. Moreover, the combination of R-WAC and cytarabine demonstrated more active anti-leukemia effects than daunorubicin and cytarabine in the AML mice model. R-WAC and cytarabine or daunorubicin and cytarabine did not exhibited decreased body weight in mice. Next, we investigated R-WAC combination with venetoclax, selective BCL-2 inhibitor, for anti-leukemia effects. R-WAC and venetoclax exhibited increased cell apoptosis against MV4-11 and KG1a cells in vitro, and more active anti-leukemia effects in xenotranplant MV4-11 mouse model than each monotherapy. These results suggest that R-WAC and combination with cytarabine or venetoclax has strong anti-leukemia effects without increased toxicities. Finally, R-WAC significantly suppressed the colony-forming capacity of bone marrow CD34 + stem/progenitor cells in refractory AML patients, indicating R-WAC depleted AML stem cells.
Conclusion
R-WAC has promising anti-leukemia effects with less toxicities compared with conventional anthracycline possibly due to the different inhibitory mechanisms of Top2 isoform (anthracycline inhibits Top2β). These results indicate that R-WAC is a promising therapeutic agent for AML and support its clinical development, especially as a combination of R-WAC with cytarabine or venetoclax.
Disclosures
Ueshima:Wakunaga Pharmaceutical Co., Ltd.: Current Employment. Kashimoto:Wakunaga Pharmaceutical Co., Ltd: Current Employment. Yamaguchi:Wakunaga Pharmaceutical Co., Ltd: Current Employment. Takashima:Wakunaga Pharmaceutical Co., Ltd: Current Employment. Sanuki:Wakunaga Pharmaceutical Co., Ltd: Current Employment. Ichinohe:Repertoite Genesis Inc.: Research Funding; Abbvie Co.: Honoraria, Research Funding; Sumitomo Pharma Co: Honoraria, Research Funding; Nippon Shinyaku Co.: Honoraria, Research Funding; Chugai: Honoraria, Research Funding; Kyowa Kirin: Research Funding; Takeda Pharmaceutical Co.: Honoraria; Nippon Kayaku Co.: Honoraria; Novartis: Honoraria; Wakunaga Pharmaceutical Co., Ltd.: Research Funding; AsahiKasei Pharma Co.: Honoraria, Research Funding; Ono Pharmaceutical Co.: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal